In the world of gelatin physical chemistry, the Isoelectric Point (pI) is a defining parameter. Whether you are in pharmaceutical capsule manufacturing, precision coating, or high-end food processing, mastering the PI is essential for ensuring product stability and premium quality.
What is the Isoelectric Point (pI) of Gelatin?
Gelatin is an amphoteric electrolyte derived from the hydrolysis of collagen. Its molecular chain contains both amino groups and carboxyl groups . The Isoelectric Point is the specific pH value at which the net charge of the gelatin molecule in solution is zero.
At this precise pH, electrostatic repulsion between molecules disappears, and the electrical charge reaches an equilibrium, causing the gelatin to exhibit unique physical properties.
1. Type A vs. Type B: The Fundamental Difference in PI
The raw materials and extraction methods dictate the pI range of the gelatin, dividing it into two primary categories:
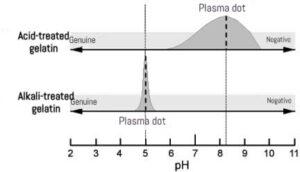
Expert Insight: The alkali treatment (Type B) causes deamidation of glutamine and asparagine, creating more carboxyl groups, which significantly lowers the pI compared to Type A.
2. Four Critical Impacts of PI on Physical Properties
At or near the PI, gelatin’s physical characteristics fluctuate dramatically, directly affecting your production efficiency:
Minimum Viscosity:
At the PI, the molecular chains are in a coiled state, resulting in the lowest viscosity and best fluidity. This is beneficial for high-concentration spray drying but should be avoided when high suspension power is required.
Minimum Swelling:
The water-absorption capacity is at its lowest at the pI, making the structure most compact. This is highly advantageous for maintaining the structural integrity of certain coatings.
Lowest Surface Tension:
This aids gelatin’s wetting and film-forming performance on various substrates—a critical metric in precision coating processes.
Gel Strength Stability:
While gel strength peaks near the pI due to molecular aggregation, being exactly at the pI may cause micro-aggregation, potentially reducing transparency.
3. Practical Industry Guidelines
A. Pharmaceuticals: Capsule Shell Stability
For hard capsule manufacturing, selecting Type B gelatin with a pI of 4.7–5.2 ensures consistent film-forming performance. If the pH fluctuates too close to the pI, it can result in uneven wall thickness or brittleness.
B. Food Industry: Clarity and Texture
Gummies & Jellies: For optimal transparency and elasticity, we recommend controlling the system pH between 6.0 and 7.0. This maintains moderate electrostatic repulsion, creating a more uniform, crystal-clear gel network.
Solubility Optimization: If you encounter dissolution issues, adjust the pH away from the pI. The resulting electrostatic repulsion helps the molecular chains expand, significantly increasing the dissolution rate.
C. Complex Interaction & Compatibility
Understanding PI guides the interaction between gelatin and other ingredients (like polysaccharides). For instance, in acidic beverages, positively charged Type A gelatin interacts better with negatively charged pectin to form stable complexes and prevent sedimentation.
4. Quality Control & Measurement
As a benchmark for purity and quality, pI is measured via:
Isoelectric Focusing (IEF): Determines both the pI value and the charge distribution.
Capillary Isoelectric Focusing: Used for high-precision R&D and strict quality audits.
Conclusion
The Isoelectric Point is more than just a scientific concept—it is the “compass” for gelatin application. By precisely managing the gap between your system’s pH and the gelatin’s pI, you can master the balance between viscosity, stability, and clarity.
Our factory provides a wide range of Type A and Type B gelatins. We can customize the PI parameters to match your specific application environment. Contact our technical team today for a detailed COA and application consultation.
