The decrease in gelatin viscosity refers to the phenomenon where the viscosity of a gelatin solution diminishes under specific conditions, such as temperature, pH, shear force, time, or chemical influence. This change may be triggered by multiple factors and significantly impacts gelatin’s application properties, including gel strength, stability, and film-forming ability.
- Temperature Increase
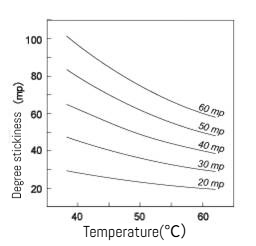
(1) Mechanism: At elevated temperatures, increased thermal motion of gelatin molecules weakens intermolecular forces (e.g., hydrogen bonds), enhancing solution fluidity and reducing viscosity.
(2) Effects: During heating, gelatin solution viscosity gradually decreases but can re-gel upon cooling (thermoreversibility). Excessively high temperatures or prolonged heating may cause molecular degradation, resulting in permanent viscosity reduction.
- pH Changes
(1) Acidic Environment: Gelatin readily undergoes hydrolysis under strongly acidic conditions (pH < 4), causing molecular chain breakage and a significant decrease in viscosity.
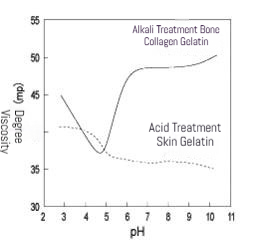
(2) Alkaline Environment: Under strongly alkaline conditions (pH > 9), gelatin molecules may undergo deamination or cross-linking reactions, leading to viscosity changes (which may increase or decrease depending on the reaction type).
(3) Neutral Environment: Gelatin exhibits maximum stability within the pH range of 5–7, with minimal viscosity variation.
- Shear Force Effects
(1) Mechanical Agitation or Pumping: High shear forces disrupt the ordered arrangement of gelatin molecules, reducing solution viscosity. This effect is typically temporary, with partial viscosity recovery possible after shear cessation.
(2) Prolonged Shearing: Sustained shear force (e.g., extended stirring) may cause molecular chain breakage, resulting in permanent viscosity reduction.
- Time Factors
(1) Aging: During storage, gelatin solutions may exhibit gradual viscosity decline due to molecular chain rearrangement or microaggregation.
(2) Enzymatic degradation: If proteases (e.g., from microbial contamination) are present in the solution, gelatin molecules undergo hydrolysis, causing rapid viscosity reduction.
- Chemical influences
(1) Salts: High concentrations of salts (e.g., NaCl, CaCl₂) may alter intermolecular forces by shielding charges or modifying solvent properties, leading to viscosity changes.
(2) Oxidizing Agents: Substances like hydrogen peroxide or sodium hypochlorite oxidize gelatin molecules, disrupting their structure and reducing viscosity.
(3) Organic Solvents: Solvents such as ethanol or acetone may decrease gelatin solubility, causing precipitation or viscosity reduction.
- Gelatin Quality and Purity
(1) Molecular Weight Distribution: Low molecular weight gelatin (e.g., Type B) inherently exhibits lower viscosity. If raw material quality or processing techniques cause molecular weight reduction, viscosity will decrease significantly.
(2) Impurity Content: Ash (inorganic salts), fat, or protein impurities may interfere with intermolecular forces and affect viscosity.
